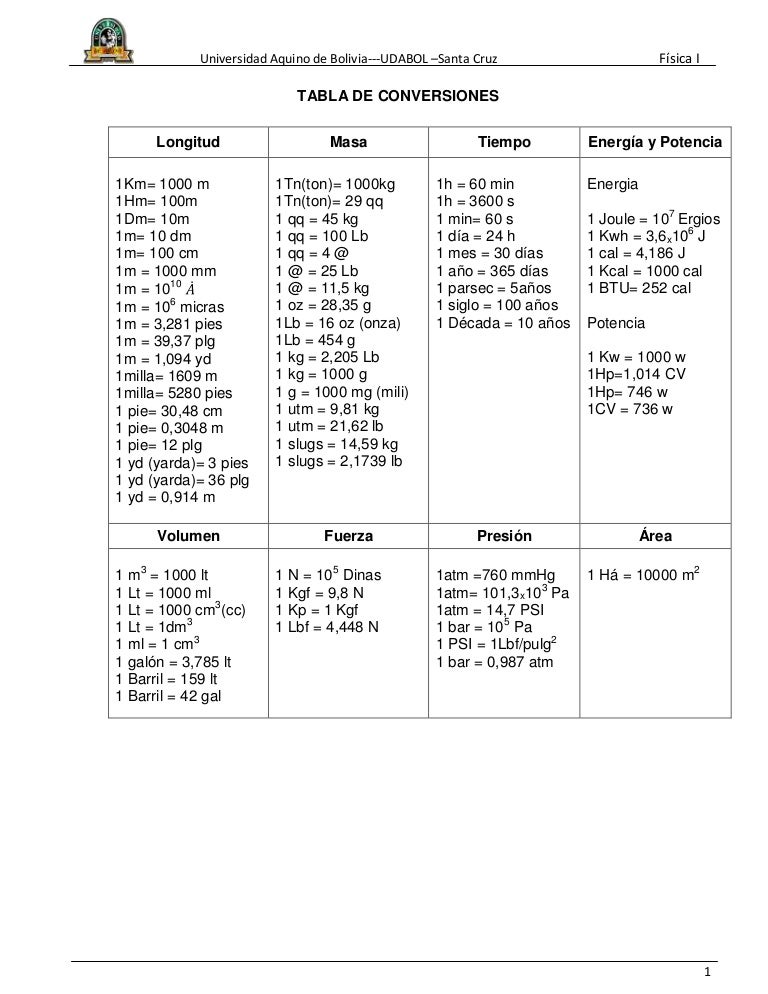
The heat Q that must be supplied or removed to change the temperature of a substance of mass m by an amount ∆T is: Q = cm∆T where c is the specific. -

Energy Changes, Reaction Rates, and Equilibrium. The capacity to do work ◦ The ability to move or change something Change in position Change in. - ppt download

Amazon.com: Nescafé Dolce Gusto - Cápsulas de café frías, 12 cápsulas (12 porciones, 1 paquete) : Comida Gourmet y Alimentos

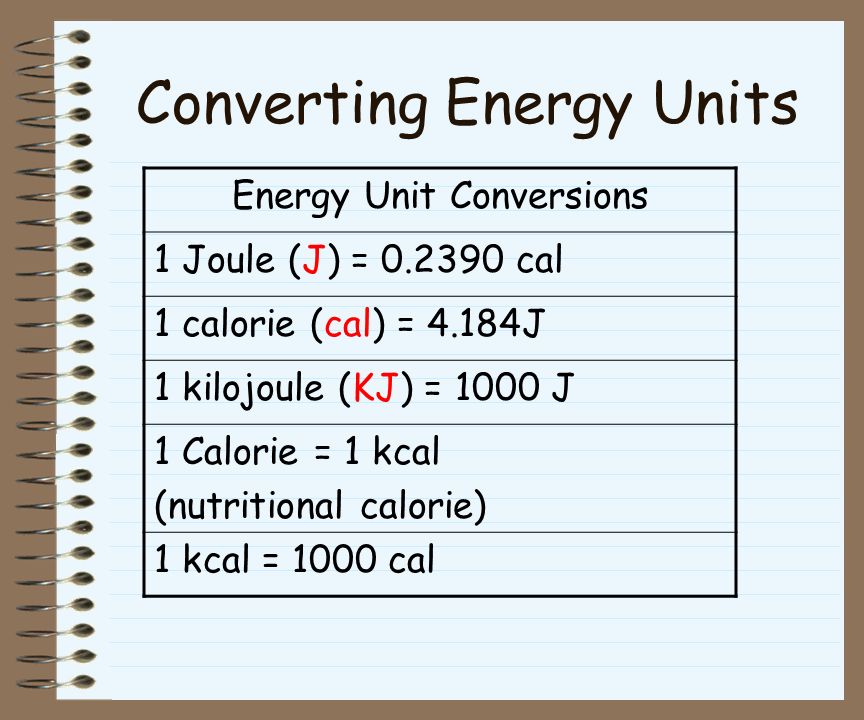
Chapter 6. Calorie One calorie expresses the quantity of heat necessary to raise the temperature of 1 g of water by 1° Celsius. Kilocalorie (kCal) - ppt download
Energy Ability to “do work” or produce a change. Forms: potential energy and kinetic energy Types: mechanical, electrical, nuclear, solar, chemical, etc. - ppt download
Solved] The average adult human burns 2.00 x 103 kcal per day in energy. What is this rate in kJ per hour ? | Course Hero
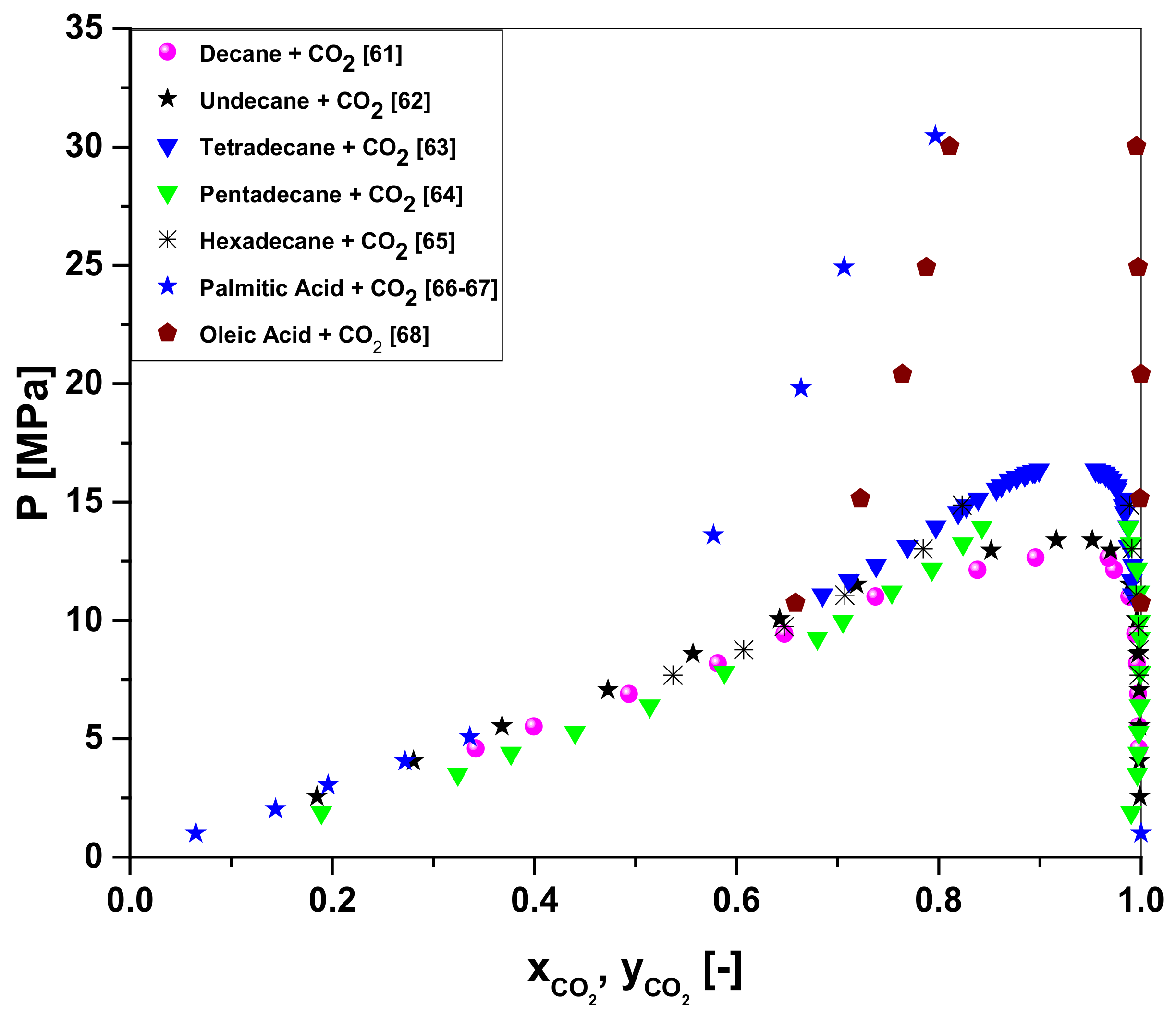
Molecules | Free Full-Text | Simulation of Organic Liquid Product Deoxygenation through Multistage Countercurrent Absorber/Stripping Using CO2 as Solvent with Aspen-HYSYS: Process Modeling and Simulation | HTML

A calorie is a unit of heat or energy and it equals about 4.2 J, where 1 J = 1 kg m^(2) s^(-2). Suppose we emp